MOOD - Molecular basis and tools for the pharmacological inhibition of the YAP-TEAD axis through the prolyl-isomerase Pin1 in cancer.
Aree / Gruppi di ricerca
Partecipanti al progetto
- Boschi Donatella (Docente)
- Lolli Marco Lucio (Ricercatore/trice)
- Martina Katia (Ricercatore/trice)
Descrizione del progetto
Project coordinator:
Prof. Giannino Del Sal, Full Professor, Università di Trieste
Members of other units:
Dott.ssa Mantovani Fiamma, Researcher, Università di Trieste
Sponsors:
MIUR - Prin 2015
Partner:
Description:
YAP is a transcriptional cofactor acting as master regulator of tissue regeneration and organ size control. Deregulation of this protein promotes cancer growth and metastasis. YAP-dependent transcriptional response is mainly mediated by the TEAD family of transcription factors, through which YAP binds to DNA. First shown as a mediator of the highly conserved Hippo signaling cascade, this complex has been recently found to be regulated by multiple upstream signals, including cell adhesion and mechanical cues and cell metabolic state, among others. This multifaceted signaling network generates on YAP a complex repertoire of post-translational modifications, however how upstream signals are integrated and regulate the assembly and transcriptional activity of the YAP-TEAD complex is still largely unknown.
Phosphorylation-dependent prolyl-isomerization catalyzed by Pin1 is a widespread and extremely efficient mechanism for transducing signal-dependent phosphorylation into conformational changes affecting the biological functions of client proteins. Pin1 is overexpressed in the majority of cancers and is essential for tumor growth and progression, acting as an amplifier of several oncogenic signaling pathways.
We have obtained solid preliminary evidence that Pin1 interacts genetically and physically with YAP and that its activity is required for YAP biological functions.
Our central hypothesis is that the phosphorylation-dependent prolyl-isomerase Pin1 acts as a key transducer of oncogenic signals to control YAP-TEAD activation and function in cancer.
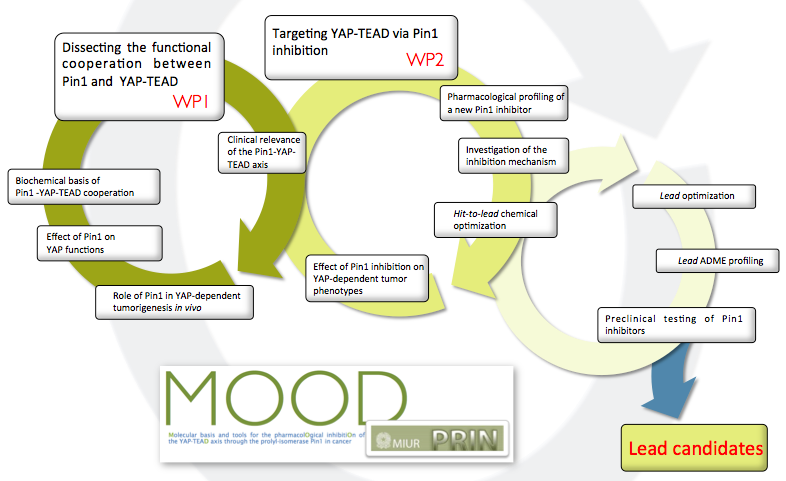
Our aims are: - To dissect the interaction between the YAP-TEAD complex and Pin1, and to understand how Pin1 impacts on the execution of YAP-dependent transcriptional program. - To establish the relevance of the Pin1-YAP-TEAD axis for cancer in cell-based, pre-clinical and clinico-pathological settings. - To block in vivo the Pin1-YAP-TEAD axis with small molecules targeting Pin1.
The results of this project will provide novel insights into the dynamics of YAP-TEAD complex assembly and function and will open the exciting therapeutic opportunity to target Pin1 as a strategy to block multiple oncogenic signals that simultaneously converge on YAP. Notably, both YAP and Pin1 have been shown to be required for cancer stem cell maintenance but dispensable for normal homeostasis. Therefore, the development of strategies aimed at inhibiting Pin1 has great medical and economic potential, being expected to display specific activity towards aggressive tumors.
Keywords:
Links: www.medsynth.unito.it




